Smart Omix – End-to-End Digital Clinical Research
Enabling real-world, end-to-end digital clinical research for independent and institutional teams: from study design to participant engagement to data analysis.
Role
Product Designer (Sole)
Team
Startup team
Year
Sep 2021 – Jul 2022
01 — Context & Opportunity
Clinical research typically means in-person studies, which are resource-heavy and inaccessible for many. Digital platforms offer more flexibility, but can make researcher-participant relationships harder to build. Smart Omix set out to change this: enabling real-world, end-to-end digital research for independent and institutional teams.
The platform has two sides: a researcher web app for designing studies, managing participants, and viewing data, and a participant mobile app for enrollment, data entry, and rewards. I joined as the sole designer in September 2021, leading up to the July 2022 launch, and continued with V2.
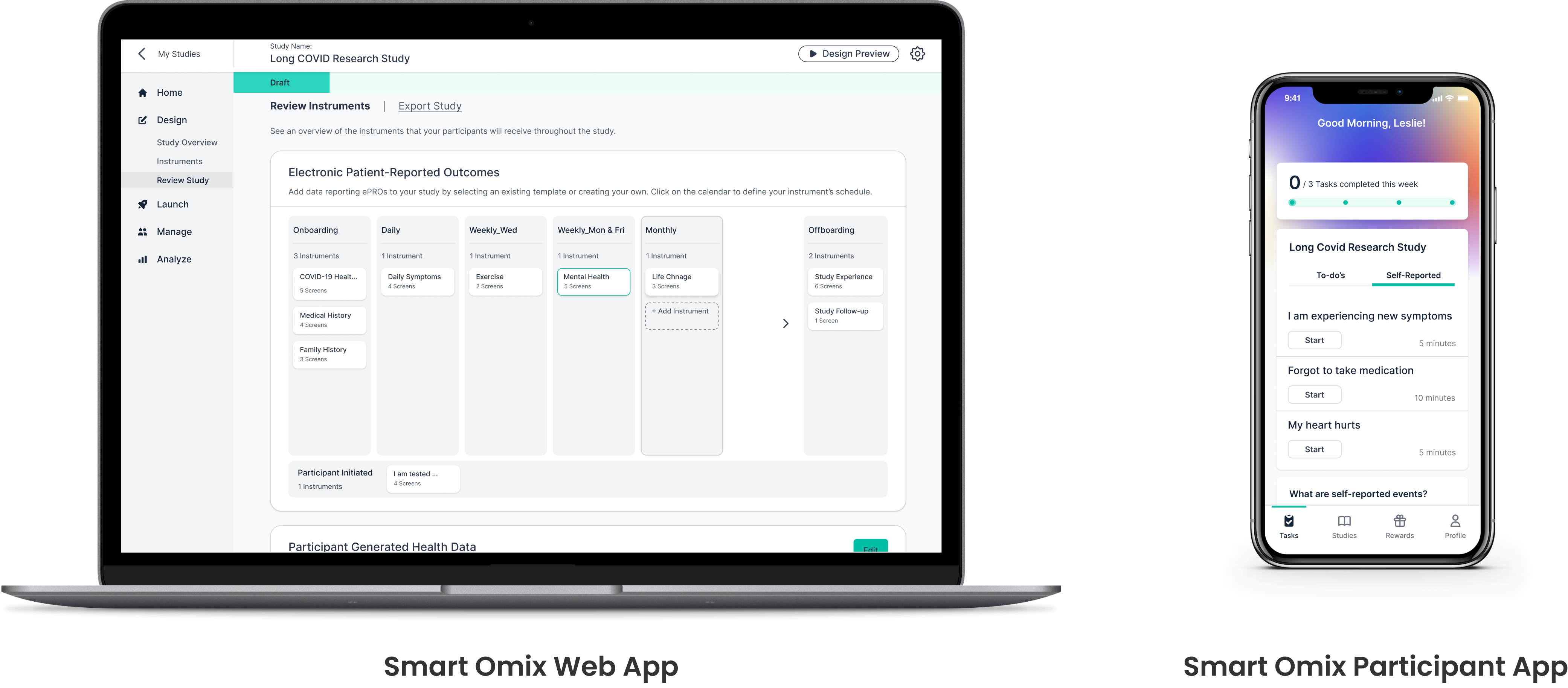
02 — My Responsibilities
I defined user stories and product features, mapped site architecture and flows, iterated designs, facilitated reviews, delivered specs to engineering, ran usability tests, and produced marketing assets.
03 — Key Challenges & Strategic Solutions
1. Platform for Complexity and Scale
Problem
Researchers' needs ranged widely: some simple studies could be managed by a single person, while complex studies often required whole teams. Each member with distinct roles, responsibilities, and goals. We needed a flexible structure that could accommodate both ends of this spectrum and everything in between.


Solution
- I designed a phase-based navigation: Design, Launch, Manage, Analyze, mirroring research stages.
- Organized studies in "organizations" with layered permissions, making access and roles scalable.
- Built modular components for each phase: simple for new users, flexible for complex teams.

2. Managing Nonlinear Study Life Cycles
Problem
Studies rarely follow a straight line; phases often overlap. For example, while a study might be locked for IRB review or launched, researchers inevitably discover changes they need to make, even after locking. We needed a nuanced system that allowed users to lock and unlock studies as needed, while automatically managing all downstream workflow adjustments to keep everything in sync and error-free.
Solution
- Built a flexible state machine to switch easily between phases
- Clarified allowed actions at each stage to minimize confusion

3. Supporting New, Data-Rich Research
Problem
Switching to digital introduced new data types and uncertainty.

Solution
- Designed the platform to educate and encourage adoption of novel data through customizable study settings
- Used a layered information architecture: summarizing at a high level, with details available on demand
- Enabled researchers to preview the participant interface, so they could visualize what participants would see and better tailor the study design
4. Accessibility, Despite Complexity
Problem
Clinical research is inherently complex, but we wanted Smart Omix to be approachable for all researchers, regardless of their experience level. Our goal was to support both novices and experts by designing an intuitive, streamlined flow. We built in educational tools and context-sensitive guidance to help those who needed it, but ensured these resources were non-intrusive and wouldn't disrupt the workflow of experienced users.
Solution
- Layered Informational UI: Added an informational layer on top of the core UI, revealing helpful content and tips through context-sensitive triggers to support users when needed.
Floating information window triggered by hover - Step-by-Step Mini Tasks: Broke complex study creation into mini tasks with stepped guidance at key moments, providing structure and direction while preventing users from getting overwhelmed.
Steps of creating an ePRO - Ghosting Future Actions: Used ghosted previews of upcoming steps and options so users could stay focused on what's next, while building awareness of the overall process.
Choose which content to ghoast based on which step user is at, to help them build expectation without being overwhelming
5. Building Transparency and Trust
Problem
Digital platforms can feel impersonal, so we focused on keeping researchers and participants connected. Traditionally, in-flight studies often behave like a black box: researchers have limited visibility and control once a study is underway. We wanted to change that by ensuring researchers could easily review progress and make necessary adjustments at any point, as well as reach out to participants if needed.
Solution
- Built in notifications and alerts so researchers could track progress and participant needs
Notify researchers of the participants needing attention - Created dashboards for actionable insight across participant groups
Dashboards allow researchers to view study data from various angles
04 — Execution & Results
Execution Principles
Strong early product architecture, a cohesive vision, and disciplined synthesis of stakeholder input were key to scaling Smart Omix.
Designed for edge cases
Built flows for critical scenarios from the start
Full-cycle ownership
Led usability testing and iteration, drove roadmap alignment
Consistency over novelty
Prioritized predictable patterns to reduce learning curve and support needs
Results
Smart Omix expanded access to clinical research, enabling greater complexity, rapid iteration, and real engagement on a platform built for scale from day one.
Reflections
- Set up for scale: A big-picture, scalable structure from the outset saves major headaches down the line and ensures smooth transitions as the product grows.
- Go beyond UI/UX: True product design means understanding domain, user pain points, compliance, engineering costs, and feasibility.
- Own the design: Stakeholder feedback is critical, but the designer must synthesize and make the final call with confidence.
- Prioritize consistency: Design consistency across both broad concepts and the smallest details matters most, especially in complex or phased products.